Terumo: FDA sets $35M fine and manufacturing restrictions for Scio Township medical device maker
(This story has been updated several times with additional information.)
Terumo Cardiovascular Systems Corp. has agreed to pay $35 million in fines and to scale back manufacturing and sales of several of its medical devices after the U.S. Food and Drug Administration uncovered "numerous" violations at the company's Ann Arbor-area headquarters.
The company signed a consent decree agreeing that it will not
manufacture or sell two heart-lung bypass systems and several other
medical devices to new customers until it makes improvements to its
manufacturing and reporting systems as required by the FDA, officials said today.
Terumo CVS — which has 435 employees at its Scio Township operation, up from 367 a year ago — can continue to sell the products to existing customers, a spokeswoman said.
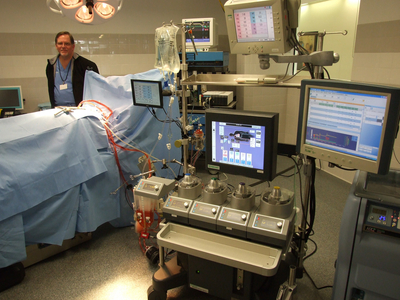
Terumo Cardiovascular Systems tests its medical devices in a simulated operating room.
Nathan Bomey | AnnArbor.com
Among the accusations is that Terumo CVS did not properly investigate or report possible problems doctors encountered with the company's medical devices.
According to FDA documents provided to AnnArbor.com, four FDA investigators who inspected the Scio Township facility in early 2010 concluded that Terumo CVS had “not implemented an adequate and effective quality system at all levels of your organization.”
The issues cited by the FDA investigators included an incident in which a patient died some time after Terumo CVS’ “soft flow arterial cannula” was removed from the patient with a blood “clot found entangled in the diffuser tip of the cannula.”
“Your firm did not document the attempts to retrieve the product from the hospital, or document the reason why no products were returned for investigation,” the Detroit-based FDA investigators concluded in their report. “The investigation conducted by your firm did not include any attempts to try and recreate the problem on the remaining inventory in stock. Only a visual inspection of the remaining stock was conducted.”
The investigators added: “The investigation did not determine if your firm’s product caused or contributed to the patient’s death.”
In response to the FDA's assertions, Terumo and the hospital that treated the patient jointly investigated the incident and found that the cannula had nothing to do with the patient's death, said Barbara Schmid, Terumo CVS' director of corporate communications.
"We are 100 percent convinced and so is the hospital that there is no relation between that cannula and that patient’s death," she said. "Cardiac surgery is a very, very common procedure and our products are used many, many times every day."
The FDA, she said, only "felt that we did not do adequate investigation into the incident. That needs to be clear."
Terumo CVS' manufacturing problems date back to warning letters FDA investigators distributed to the company in 2004 and 2006.
The company shares its sprawling 55-acre Scio Township campus with Terumo Heart. The sister companies are subsidiaries of $3.88 billion Japan-based Terumo Corp.
The firm said in a news release this morning that it would "remain an important member of the southeast Michigan business and life science communities" and that it would "continue contributing to the economy (in Ann Arbor) by providing high-quality jobs, hiring local suppliers and supporting community activities."
Schmid said job cuts were unlikely.
"We currently believe that we'll get through the work plan for the consent decree without the loss of jobs," she said in an interview. "We have totally committed to staying in the Ann Arbor area through all of this in case anyone thought there was a chance that we would be leaving the area or diminishing our capacity in the area."
For now, customers using Terumo products — that is, hospitals, health care systems and patients — can continue using them because the FDA is concerned that pulling them off the market would cause disruptions. Terumo says customers use its products in "1,000 life-saving procedures ever day."
Terumo CVS CEO Mark Sutter said in a statement that the firm would fix the manufacturing problems.
“Terumo CVS is committed to fully addressing all of the FDA’s concerns," he said. "Over the past year, Terumo CVS had already begun implementing a significant quality system initiative that will create systemic, sustainable improvements in its quality systems. Terumo CVS has always been, and will remain, committed to those in the cardiac surgery community who use our products.
Terumo CVS products impacted by FDA investigation
The consent decree between the FDA and Terumo Cardiovascular Systems restricts the manufacturing and sale of the following devices to existing customers only, until the company fixes "numerous" problems at its Scio Township manufacturing facility.
- Terumo Advanced Perfusion System 1
- Sarns Modular Perfusion System 8000
- Sarns Centrifugal System
- HX2 Temperature Management System
- Sarns TCMII Cooling and Heating System
- CDI 500 Blood Parameter Monitoring System
- Sarns Sternal Saw II System and Replacement Blades
- T-Link Data Management Systems (software upgrades)
- Cannulae for cardiopulmonary bypass
- Cannulae for cardioplegia delivery
- Vents, suckers, dilators, connectors and reducers.
"Our ultimate goal during the consent decree is minimizing the inconvenience or disruption to our customers."
The FDA said investigators visiting Terumo CVS from January 2010 to March 2010 found "numerous" violations, including "deficiencies in processes for corrective and preventive action, nonconforming product, complaints, purchasing, process validation, design controls, and adverse event reporting."
An FDA spokeswoman was not immediately available to comment.
“Medical device manufacturers must comply with the FDA’s current Good Manufacturing Practice and Medical Device Reporting requirements, as this action demonstrates,” said Steven Silverman, director of the Office of Compliance in the FDA’s Center for Devices and Radiological Health, in a statement. “These requirements protect patients by assuring that medical devices are safe, effective, and high quality.”
Terumo had previously been held up by the Michigan Economic Development Corp., Ann Arbor SPARK and industry leaders as an example of the Ann Arbor region's medical device prowess. Terumo's two Scio Township divisions in late 2009 completed a $3.6 million expansion that included 60 new jobs over an 18-month stretch.
“Terumo is kind of a secret in a way,” Sutter said at a press conference in March 2010. “We’ve been here for many years and we don’t talk about ourselves a lot. We could not exist without the great, great support we have in this community and in the state.”
In 2008, Terumo received a 7-year, $855,000 tax credit from the MEDC's Michigan Economic Growth Authority board to support its expansion and hiring.
Schmid said the company has been doing "a lot of hiring" to address the FDA's quality concerns.
"We certainly have put a lot of work into our quality systems," she said. "We have a lot of examples of that, and we made a lot of progress, but the FDA believes the actions we took were not sufficient to bring our systems into compliance with their (policies)."
Terumo CVS is aiming to complete the FDA's required manufacturing and reporting improvements within two years.
Schmid said the list of Terumo products that are affected by the FDA investigation represent "a relatively small amount of our overall revenue."
Terumo's Ann Arbor roots run deep. Longtime Ann Arbor entrepreneur and medical device inventor Dick Sarns launched Sarns Inc. in the 1960s and sold it to 3M Corp. in the 1980s. Terumo's parent company acquired Sarns-3M in 1999, forming the new Terumo Cardiovascular Systems subsidiary.
In 2008, the company consolidated manufacturing operations from Mexico, Indiana and California and shifted work to its Scio Township division, choosing the Ann Arbor region as its corporate headquarters.
Contact AnnArbor.com's Nathan Bomey at (734) 623-2587 or nathanbomey@annarbor.com. You can also follow him on Twitter or subscribe to AnnArbor.com's newsletters.


Comments
Michisbest
Wed, Mar 23, 2011 : 6:03 p.m.
Many of these comments are so far off they prove the old adage "better to be quiet and thought a fool than open your mouth and remove all doubt" just plain speaking.
jcj
Wed, Mar 23, 2011 : 1:31 p.m.
@ fredric & RLB Very refreshing to see two comments by persons that do not appear to have an agenda other than being honest about what was written. Thanks
fredric
Wed, Mar 23, 2011 : 12:46 a.m.
I am here writing this note because one of these systems was used on me during a 4 quad by-pass back in 2000. I feel lucky to have had the product available at the U of M medical center. What is really interesting, I worked for DIck Sarns when he owned the company back in the late 60's, when I got out of the service and returned to Ann Arbor. They had a very solid program in place back they for quality control and also a person with experience in operating this equipment went to the Hospitals with the equipment, to insure that the Hospital staff was properly trained in its operation, Most times when a equipment fails, you will find that is the result of an operator error!. None of us know the reason why the person passed, but many 100,000 's of thousands are probably living a normal life because of this equipment and we should be proud that it was built right here in our home town of Ann Arbor ! I for one , sure and thankful !!!
HJK
Tue, Mar 22, 2011 : 8:39 p.m.
"Our ultimate goal during the consent decree is minimizing the inconvenience or disruption to our customers." His ultimate goal shouldn't be about marketing and selling products. The goal should be correting the quality issues, so the end user can "by assuring that medical devices are safe, effective, and high quality." If they got a warning letter starting in 2004; they had 7 years to clean up their quality issues. They may have invested a lot of time and money on their quality, but it looks like it wasn't a good investment.
Nathan Bomey
Tue, Mar 22, 2011 : 7:35 p.m.
I've updated the story with details of Terumo's response to the FDA's assertions that the company did not properly investigate the death of a patient after use of a cannula during a heart procedure: >>In response to the FDA's assertions, Terumo and the hospital that treated the patient jointly investigated the incident and found that the cannula had nothing to do with the patient's death, said Barbara Schmid, Terumo CVS' director of corporate communications. >>"We are 100 percent convinced and so is the hospital that there is no relation between that cannula and that patient's death," she said. "Cardiac surgery is a very, very common procedure and our products are used many many times every day." >>The FDA, she said, only "felt that we did not do adequate investigation into the incident. That needs to be clear."
RLB
Tue, Mar 22, 2011 : 7:17 p.m.
A clot at the end of a cannula or catheter in a patient is not uncommon. The fact that a patient died after getting a clot at the end of one of Terumo's cannula doesn't necessarily mean that the product was defective. The problem the FDA had is that Terumo didn't investigate the death thoroughly enough. They either didn't follow their own internal procedures or their internal procedures weren't thorough enough to meet FDA requirements. That's what they're getting in trouble for. The FDA probably then had other findings during their audit last year that they got dinged on: "deficiencies in processes for corrective and preventive action, nonconforming product, complaints, purchasing, process validation, design controls, and adverse event reporting." The key word there is "processes" - basically what Terumo's quality system had documented for what requirements need to be met in each of those different areas.
Trepang674
Tue, Mar 22, 2011 : 6:42 p.m.
I wonder if they implemented a proper Disciplined Problem Solving Process to find the root cause...If so, than did they have to have the machine returned and evaluated...What caused the clot? The machine or the process. If the FDA fined them and they agreed to pay, does that mean they were negligent or did they get pressured by the FDA to plead.? Where again does Rick Snyder fit into this engineering problem? Lordy...any way to get a weary gripe in.
clownfish
Tue, Mar 22, 2011 : 6:05 p.m.
Not sure is this is due to the reporting of statements issued by Terumo, but.. why comment on possible job loss, and not life loss via defective products? good work by the FDA on this one! Keep it up!
noreaster
Tue, Mar 22, 2011 : 7:28 p.m.
"good work FDA"??? It took the FDA a whole year to report on a single death plus some site inspections that together probably lasted no more than a week. In the mean time have any more died as a consequence of these violations? Thanks FDA, nothing like a prompt evaluation - NOT! Looks to me the FDA staff is perhaps a bit gutted. Oh wait, that likely happened under the Republican leadership of G. W. Bush. Thanks for looking out for us...
say it plain
Tue, Mar 22, 2011 : 6:14 p.m.
Exactly! That the focus here is jobs is perhaps not surprising, but man even for a business report section that wants to play constant scorekeeper and chearleader for companies employing locally, this is a depressing story! It certainly served to make me feel very unhappy about them; who cares how many people they employ. Indeed, it makes me wonder about those who are employed by the firm...why were stricter quality control operations not in place, and what is 'wrong' with the product they are sending out the door?! This isn't like a debate about who puts out a car cabin with tighter bodies, gee, this is about whether a machine used in delicate surgeries might have unacceptable engineering/manufacturing. "Local" economic impact consideration is so, unseemly...
say it plain
Tue, Mar 22, 2011 : 5:39 p.m.
They should definitely be exempt from all further state subsidies, because they have clearly violated "best practices". Or, in Snyder's accounting, does a business which fails to take *very* seriously the possibility that a design flaw in their vitally important product could cause *deaths* so as to avoid losing some bottom-line profit qualify as engaged in "best practices"?! They didn't manage to avoid fines and restrictions, so they must not have hired the "best" corporate lawyers, too bad for them!
Tailgate Jim
Tue, Mar 22, 2011 : 5:16 p.m.
I maybe wrong but I thought Granholm was Gov. in 2008
AA
Tue, Mar 22, 2011 : 4:46 p.m.
Now can they still get the $nyder Buck$ to help pay the fine. They are a local company that needs money now more than ever before